Indian Regulatory Unveil the Risk Class of Recently Notified Medical Devices

In an effort to develop a more
dignified system of registration, India is implementing some new rules for
medical devices that are sold and distributed in the country. The new medical devices rule 2017 contains
regulations regarding the use of risk-based classification of medical devices
as well as the in-vitro devices along with the execution of unique device
identification requirements. In addition to that, the manufacturers of the
medical devices are subjected to manufacturing quality audits that are done by
notified bodies specially authorized to do such audits by the government. Some
of the rules also concern fee change depending on the new system.
The new regulation favours issuing
license that is valid for three years rather than one year. The companies that
possess devices that are already registered for selling in India will be given
a transition period during which they can comply with the new rules. The new Medical
Device Registration India rules by the Indian regulatory authorities also unveiled the
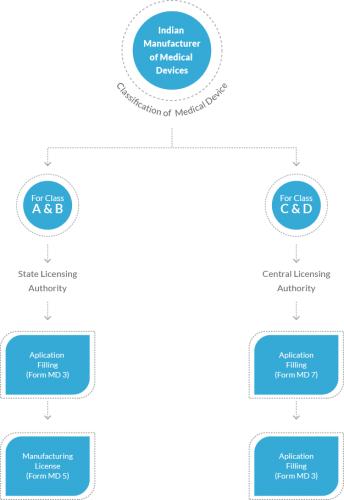
risk class of medical devices. Before knowing about the details, you must know
whether regulator classification matters or not.
·
The medical classification assists in the easy determination
of what procedures you should follow before selling product.
·
It helps in establishing what is required, especially at the
time of development of the product as well as design controls.
·
It is one of the vital components for the determination of
the amount of money required for bringing the product to the market and lends
you some idea about the involved duration.
Keeping this in view and for safety
concerns, the Indian Central Drugs Standard Control Organization has set a risk
classification of medical products that have been scheduled for notification
and regulations starting from April 2020.
The notice directs to devices kinds,
and all of them will be assigned as moderate to moderate-high class B and class
C risk classifications.
·
CT scan equipment (Class C)
·
MRI equipment (Class C)
·
Defibrillators (Class C)
·
Dialysis machines (Class C)
·
PET equipment (Class C)
·
X-ray machines (Class C)
·
Bone marrow cell separators (Class B)
·
Nebulizers (Class C)
·
Blood pressure monitoring devices (Class B)
·
Digital thermometers (Class B)
·
Glucometers (Class C IVD)
·
Organ preservation solutions (Class C)
This classification as unveiled by
Indian regulatory will help the Indian medical device market registrants, as
well as their in-country representatives to obtain a good idea of problems
related to cost, clinical data needs and import licensing authority while they
sell their products in the Indian market.







Comments